Biodiesel is an alternative fuel to conventional fossil ones. The EU policies of boosting biodiesel have achieved its implementation in the transport fuels market and increasingly its sustainable nature is being taken into account. Thus, up to 86 % of the biofuels used in the EU in 2013 achieved the certificate of sustainability, in accordance with the Community norms.
Chemical engineer Ines Reyero Zaragoza proposed the substitution of methanol by ethanol for the production of biodiesel and the use of a heterogeneous catalyst, which will "result in a reduction of costs and in the environmental impact associated with the production of this biofuel." One of the novel contributions of her research has been the synthesis and identification of a new compound the characteristics of which make it "a very good candidate for developing heterogeneous catalysts."
Ms. Reyero's thesis, "Advances in biodiesel production: ethanolysis and new heterogeneous catalysts," was directed by Professor Luis Gandía Pascual and Professor María Cruz Arzamendi Manterola, from the Department of Applied Chemistry at the UPNA, and was awarded an Outstanding with Honours PhD.
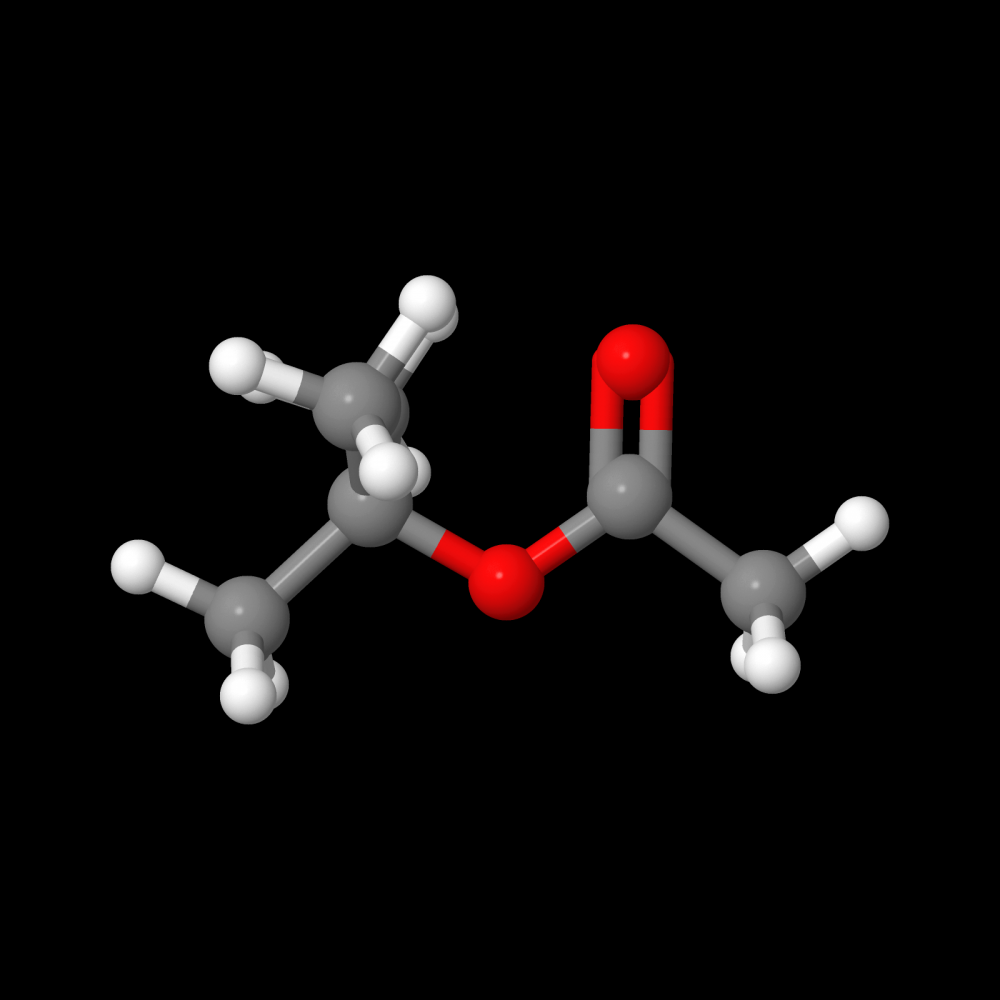
Dr. Reyero explains that, in chemical terms, biodiesel production is based on a reaction known as transesterification. "Vegetable oils are made to react with an alcohol, usually methanol, producing biodiesel and glycerine, a secondary product that has varied applications once purified." For the reaction to take place a suitable catalyst is needed. "Conventional industrial processes normally use catalysts that are labelled homogeneous because they are soluble in one of the reagents, specifically in alcohol."
In this research ethanol was used instead of methanol. The latter is made from fossil-origin raw materials, while ethanol can be obtained from renewable resources (and is thus known as bioethanol). "We have shown that biodiesel synthesis reactions have very different behaviour depending on what kind of alcohol is used. For example, while with methanol the system is biphasic, using ethanol involves a homogeneous reaction medium and single liquid phase."
The practical consequences of these variations are relevant. On the one hand, on using ethanol the speed of reaction is very much greater and, thus, there is a greater capacity of production of the reactors. "Nevertheless, on the other hand, the homogeneous character of the reaction complicates the separation of the products and favours the formation of soaps, which contaminate these products, especially glycerine."
The author of the PhD explains why heterogeneous catalysts are used instead of homogeneous: "the homogeneous ones currently employed are highly effective, but they cannot be reused and they contaminate the products, and so costly separation and purification stages are needed to obtain biodiesel and glycerine in sufficient quantities to be commercially viable. Moreover, all these processes produce large amounts of waste water that also need to be suitably treated."
One novel contribution in the course of this research has been the synthesis and identification of a new compound - calcium glycerolate - "which notably surpasses calcium oxide and glyceroxide in stability and makes it a very good candidate for the development of heterogeneous catalysts for the synthesis of biodiesel."
Ms. Reyero has also used structured catalysts which, in the active phase, are deposited on ceramic or metallic structures. This enables the passage of the reaction medium and facilitates the transformation of the reagents into products - biodiesel and glycerine in this case. Thus, the separation of the catalyst from the reaction medium is avoided, and its reuse facilitated. "The use of structured catalysts is highly novel in the field of biodiesel production and has meant an important technological advance" pointed out the researcher, "and so, in this sense, this thesis can be considered pioneering."
Source: Basque Research