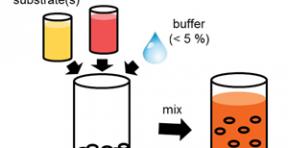
Biocatalytic "cocktail" of milled cells can solve the problems of application the enzymes in organic synthesis, related with difficulties associated with extraction of these biocatalysts and their poor solubility.

It's supposed that cell membrane helps to stabilize enzyme in organic solvent.
Enzymes are excellent catalysts to observe chiral compounds. Often during the reaction under mild conditions in the one-pot mode there can be used multiple catalysts that allows to establish multi-step syntheses, once loaded it with reagents. However, if the enzymes are used as catalysts, these biocatalysts should be extracted and purified, it's also often necessary to add another expensive cofactors in this system.
The problems also arise from solubility - the highest enzyme activity performs in buffer solutions, which pH values correspond to the physiological values of native state of the enzyme, however, since most of the substrates of these enzymes are hydrophobic, it becomes not possible to obtain a high concentration of the substrate in an aqueous buffer, whereby the rate of reaction is considerably reduced and, consequently, its efficiency.
Dörte Rother and Andre Jakoblinnert found the approach to solve this problem. Researchers had two step biocatalytic reaction taking as a catalyst lyophilized cells of recombinant Escherichia coli, containing these enzymes instead of purified and isolated enzymes.
To make such a reaction there may be also used an organic solvent, which can increase the substrate concentration in the reaction mixture. Previously it was reported only about four examples of biotransformation, where all of the reactions were a single-stage processes.

To carry out a two-stage cascade reaction catalyzed by lyophilized cells, there is no need to add enzyme cofactors into reaction mixture.
Nicholas Turner, a specialist in biological catalysts at University of Manchester explains what is the advantage of the new strategy. He notes that Rother and Jakoblinnert succeeded to use carboligase and oxidoreductase at the same time to convert inexpensive raw materials in optically pure 1-phenyl-propane-1 ,2-diol.
Vicinal diols like this compound are used for manufacturing pharmacologically active substances, such as diltiazem (antiarrhythmic antihypertensive calcium channel blocker), and also to produce compounds used in agriculture. Developed process that is characterized by high stereoselectivity (ee / de> 99%) suggests the reactions catalyzed by lyophilized cells as a perspective new strategy for the synthesis.
Hopefully, the application of the cells as catalyst can considerably make the process easier and decrease its risks of environmental pollution, however, to proceed this it's necessary to determine E-factors, values that shows the amount of waste generated by one unit of desired product's mass.